What Is GLP-3 (RT) + Cagrilintide?
GLP-3 (RT) + Cagrilintide is a research blend that combines two compounds studied in metabolic research:
- GLP-3 (RT)
• Cagrilintide
Researchers study combinations like this to understand how different metabolic signaling systems interact.
In experimental models, scientists investigate how these compounds participate in biological signaling related to metabolism and energy balance.
This product is supplied for laboratory research purposes only.
Laboratory Research Study Areas
Researchers study these compounds in experimental models related to:
GLP-3 (RT)
- GLP-1 receptor signaling
• metabolic regulation pathways
• energy balance mechanisms
Cagrilintide
- amylin receptor signaling
• metabolic hormone signaling
• interaction between metabolic regulatory systems
All studies are conducted in controlled laboratory environments.
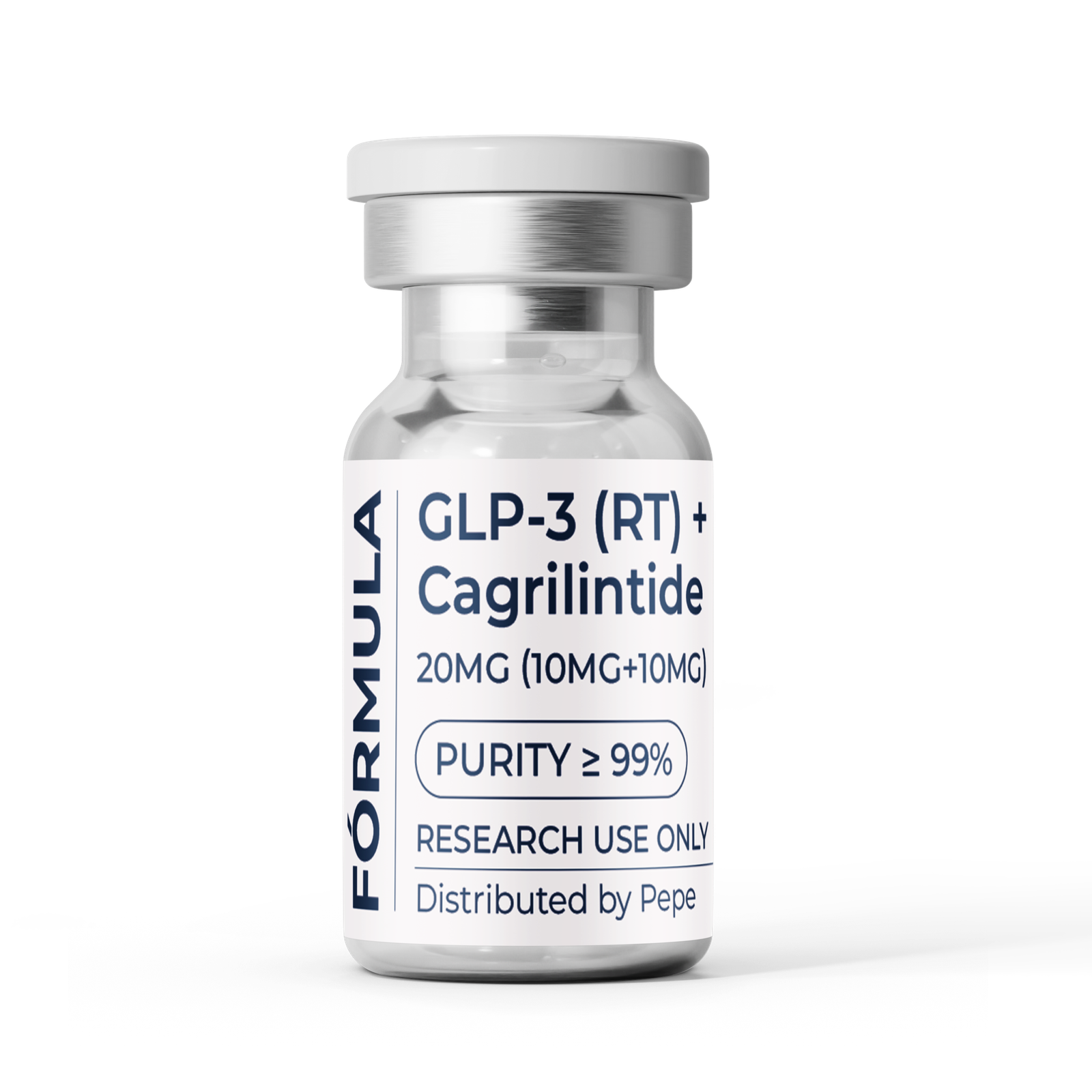
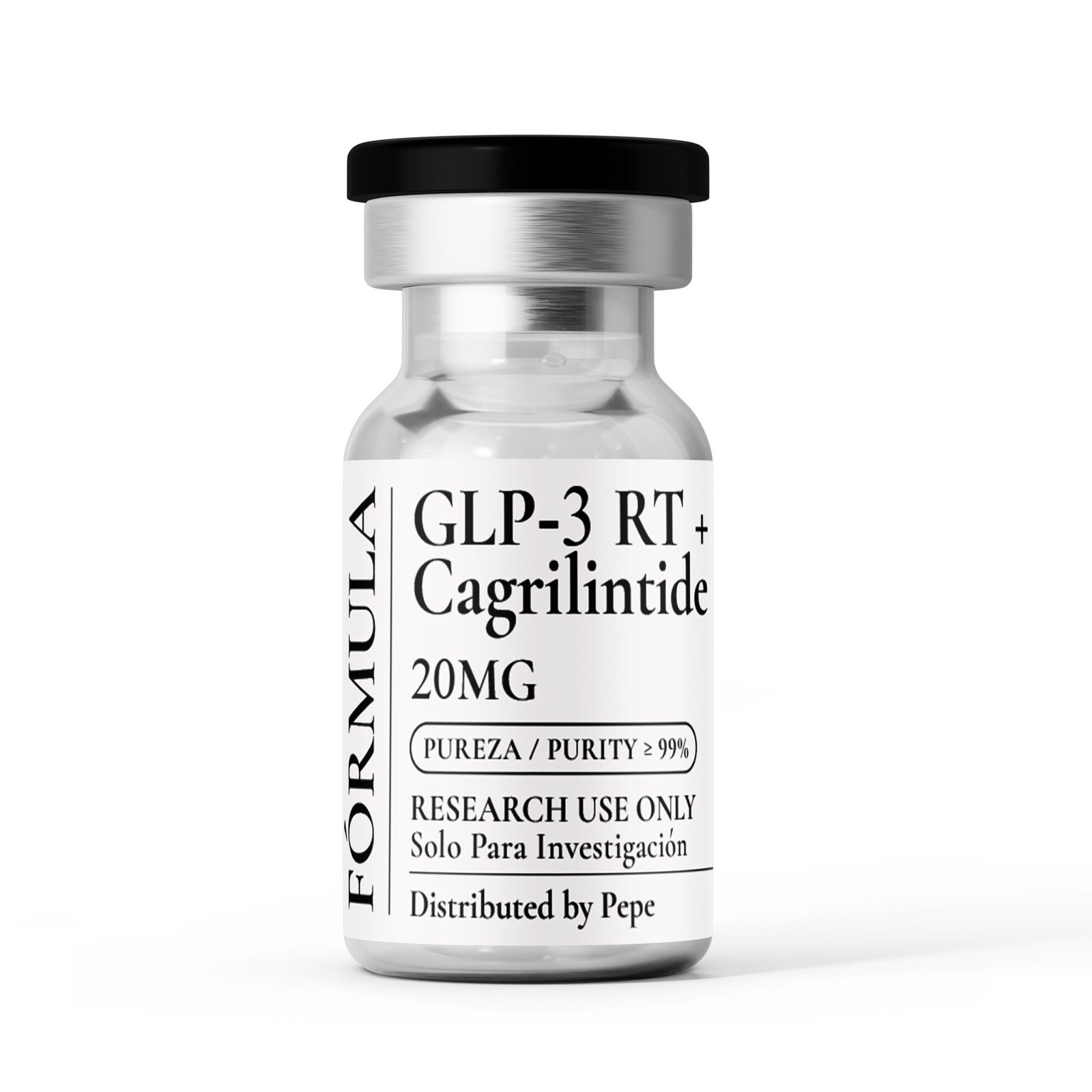
Active Ingredients
(Per vial — 20 mg total)
GLP-3 (RT) — 10 mg
Cagrilintide — 10 mg
References
Holst JJ. (2007)
The physiology of glucagon-like peptide-1
https://pubmed.ncbi.nlm.nih.gov/17344288/
Lau J et al. (2015)
Discovery of the once-weekly GLP-1 analogue semaglutide
https://pubmed.ncbi.nlm.nih.gov/25657022/
Lutz TA. (2010)
The role of amylin in the control of energy balance
https://pubmed.ncbi.nlm.nih.gov/20507899/
Frias JP et al. (2021)
Cagrilintide and semaglutide combination for metabolic research
https://pubmed.ncbi.nlm.nih.gov/33891373/
Product Specifications
Total Content: 20 mg
Composition:
GLP-3 (RT) — 10 mg
Cagrilintide — 10 mg
Form: Lyophilized powder blend
Classification: Metabolic research blend
Research Use Statement
For laboratory research purposes only.
Not for human or veterinary use.
Not intended to diagnose, treat, cure, or prevent any disease.
Distributed by Pepe

Reviews
There are no reviews yet.